Dr. Hendrick L. Bethlem
NEWS
WE ARE LOOKING FOR A PHD STUDENT FOR BUILDING
"A CRYOGENIC SOURCE OF BAF MOLECULES
- 3-7-2017: Our paper on studying cold collisions in a molecular synchrotron is out;
see arXiv1706.10236.
- 22-4-2017: The dutch Journal of Physics features a story on our fountain (in dutch);
see NTvN-mei2017.
- 7-4-2017 Aernout van der Poel wins Young Speaker Contest at Fysica 2017;
see News_archive.
- 19-01-2017: Cunfeng Cheng wins poster prize at Physics@Veldhoven;
see Poster Prize Ceremony.
- 13-12-2016: Our paper on the fountain is published;
see Cheng et al. PRL117_253201(2016).
- 15-11-2016: Our programme on
"physics beyond the standard model with cold molecules" is funded by NWO.
- 14-11-2016: Volkskrant features a story on our fountain (in dutch);
see volkskrant 14-11-2016.
Research
A molecular fountain
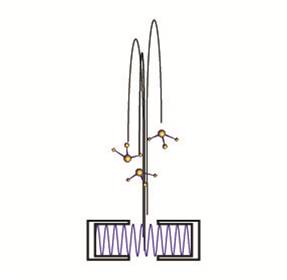 The resolution of any spectroscopic or interferometric experiment is ultimately limited by the total time a particle is interrogated. We have recently demonstrated the first molecular fountain, a development
which permits hitherto unattainably long interrogation times with molecules. In our experiments,
ammonia molecules are decelerated and cooled using electric fields, launched upwards with a velocity between 1.4 and 1.9 m/s and observed as they fall back under gravity. A combination of
quadrupole lenses and bunching elements is used to shape the beam such that it has a large position
spread and a small velocity spread (corresponding to a transverse temperature of <10μK and a
longitudinal temperature of <1μK) when the molecules are in free fall, while being strongly focused
at the detection region. The molecules are in free fall for up to 266 milliseconds, making it possible
to perform sub-Hz measurements in molecular systems and paving the way for stringent tests of
fundamental physics theories.
The resolution of any spectroscopic or interferometric experiment is ultimately limited by the total time a particle is interrogated. We have recently demonstrated the first molecular fountain, a development
which permits hitherto unattainably long interrogation times with molecules. In our experiments,
ammonia molecules are decelerated and cooled using electric fields, launched upwards with a velocity between 1.4 and 1.9 m/s and observed as they fall back under gravity. A combination of
quadrupole lenses and bunching elements is used to shape the beam such that it has a large position
spread and a small velocity spread (corresponding to a transverse temperature of <10μK and a
longitudinal temperature of <1μK) when the molecules are in free fall, while being strongly focused
at the detection region. The molecules are in free fall for up to 266 milliseconds, making it possible
to perform sub-Hz measurements in molecular systems and paving the way for stringent tests of
fundamental physics theories.
A molecular synchrotron
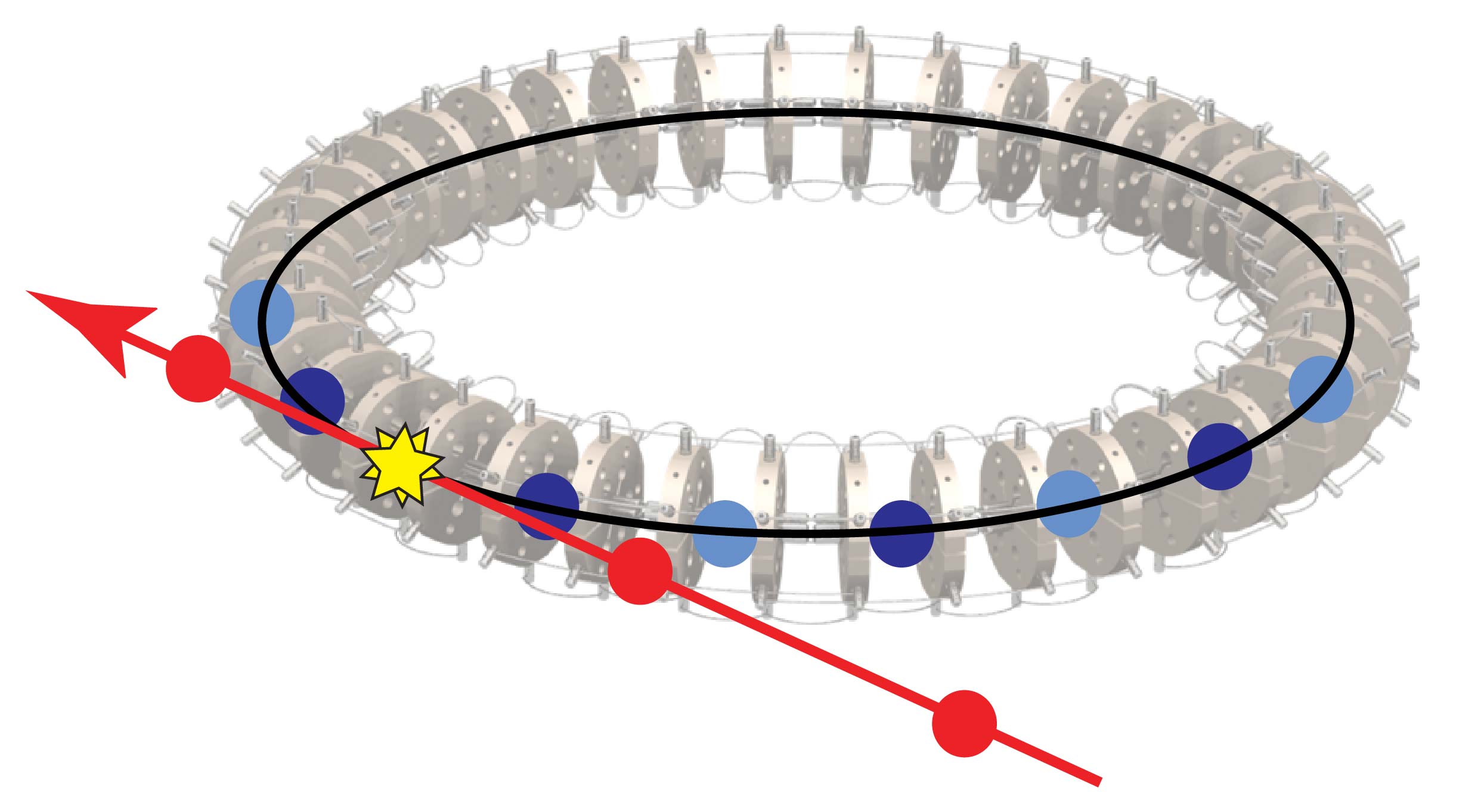 Rather than trapping particles in a trap, as is common in atomic physics,
particles can also be stored in rings. We have developed a synchrotron for
low-energy neutral molecules composed of 40 hexapoles arranged in a circle.
By switching the voltages applied to the hexapoles every time the molecules
pass through a gap, the molecules are kept in a tight bunch. We use this synchrotron to study collisions between stored ND3 molecules and beams of argon atoms. The advantage of using molecules stored in a synchrotron is two-fold: (i) The collision partners move in the same direction as the stored molecules, resulting in a low collision energy (down to 10cm-1); (ii) by storing molecules many roundtrips, the sensitivity to collisions is greatly enhanced.
Rather than trapping particles in a trap, as is common in atomic physics,
particles can also be stored in rings. We have developed a synchrotron for
low-energy neutral molecules composed of 40 hexapoles arranged in a circle.
By switching the voltages applied to the hexapoles every time the molecules
pass through a gap, the molecules are kept in a tight bunch. We use this synchrotron to study collisions between stored ND3 molecules and beams of argon atoms. The advantage of using molecules stored in a synchrotron is two-fold: (i) The collision partners move in the same direction as the stored molecules, resulting in a low collision energy (down to 10cm-1); (ii) by storing molecules many roundtrips, the sensitivity to collisions is greatly enhanced.
Test of the time variation of Mp/Me in methanol
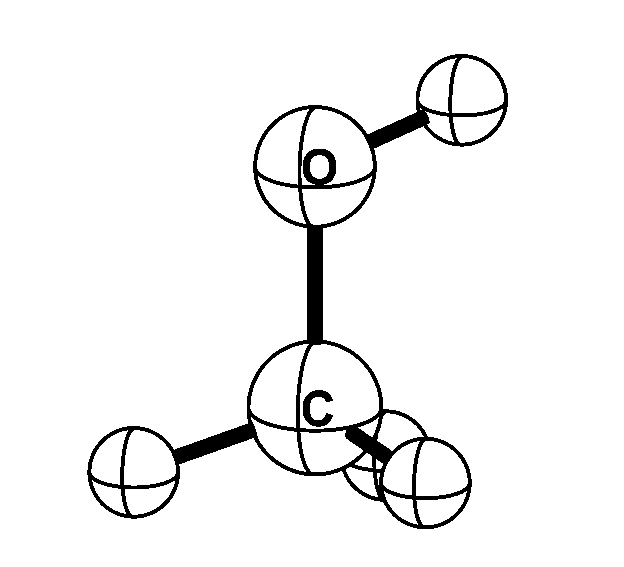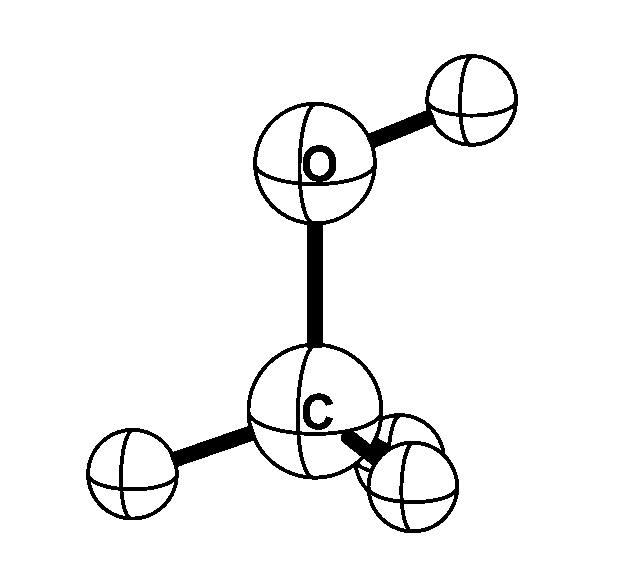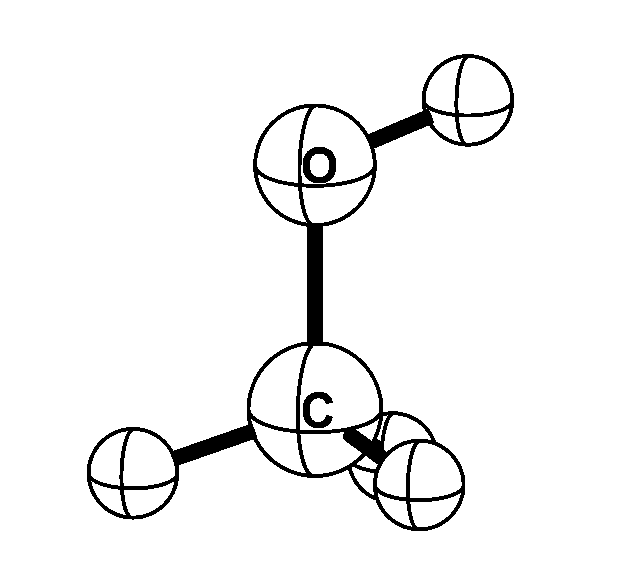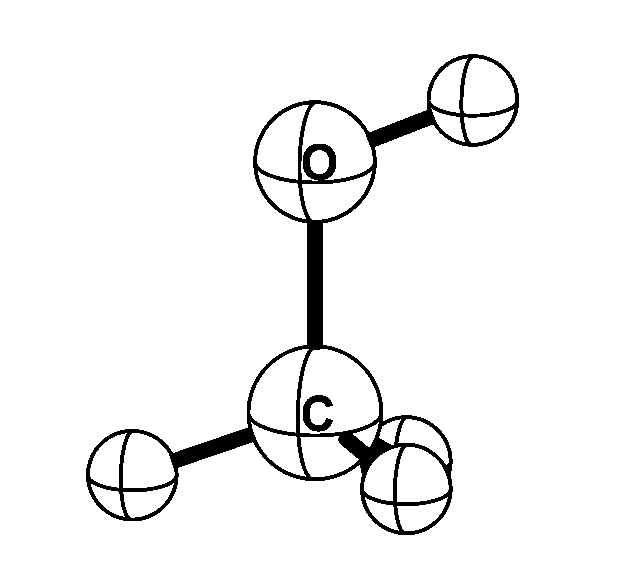 Methyl alcohol is one of the simplest molecules that exhibits internal rotation; the methyl
(CH3) group rotates with respect to the alcohol (OH) group. In addition, the molecule rotates as a whole. Microwave
transitions that convert the internal rotation to overall rotation -- and vice versa --
are very sensitive to the proton-to-electron mass ratio. When the proton-to-electron mass ratio changes by a
certain fraction, the resulting fractional frequency change in methyl alcohol is up to 50 times this fraction.
This is an order of magnitude larger than the transitions used so far in searches for possible spatial of
temporal variations of the proton-to-electron mass ratio.
Methyl alcohol is one of the simplest molecules that exhibits internal rotation; the methyl
(CH3) group rotates with respect to the alcohol (OH) group. In addition, the molecule rotates as a whole. Microwave
transitions that convert the internal rotation to overall rotation -- and vice versa --
are very sensitive to the proton-to-electron mass ratio. When the proton-to-electron mass ratio changes by a
certain fraction, the resulting fractional frequency change in methyl alcohol is up to 50 times this fraction.
This is an order of magnitude larger than the transitions used so far in searches for possible spatial of
temporal variations of the proton-to-electron mass ratio.
List of publications and theses from my group
Links
Section Atomic, Molecular and Laser Physics
Department of Physics
LaserLab
Faculty of Sciences
|


 Address
Address The resolution of any spectroscopic or interferometric experiment is ultimately limited by the total time a particle is interrogated. We have recently demonstrated the first molecular fountain, a development
which permits hitherto unattainably long interrogation times with molecules. In our experiments,
ammonia molecules are decelerated and cooled using electric fields, launched upwards with a velocity between 1.4 and 1.9 m/s and observed as they fall back under gravity. A combination of
quadrupole lenses and bunching elements is used to shape the beam such that it has a large position
spread and a small velocity spread (corresponding to a transverse temperature of <10μK and a
longitudinal temperature of <1μK) when the molecules are in free fall, while being strongly focused
at the detection region. The molecules are in free fall for up to 266 milliseconds, making it possible
to perform sub-Hz measurements in molecular systems and paving the way for stringent tests of
fundamental physics theories.
The resolution of any spectroscopic or interferometric experiment is ultimately limited by the total time a particle is interrogated. We have recently demonstrated the first molecular fountain, a development
which permits hitherto unattainably long interrogation times with molecules. In our experiments,
ammonia molecules are decelerated and cooled using electric fields, launched upwards with a velocity between 1.4 and 1.9 m/s and observed as they fall back under gravity. A combination of
quadrupole lenses and bunching elements is used to shape the beam such that it has a large position
spread and a small velocity spread (corresponding to a transverse temperature of <10μK and a
longitudinal temperature of <1μK) when the molecules are in free fall, while being strongly focused
at the detection region. The molecules are in free fall for up to 266 milliseconds, making it possible
to perform sub-Hz measurements in molecular systems and paving the way for stringent tests of
fundamental physics theories. Rather than trapping particles in a trap, as is common in atomic physics,
particles can also be stored in rings. We have developed a synchrotron for
low-energy neutral molecules composed of 40 hexapoles arranged in a circle.
By switching the voltages applied to the hexapoles every time the molecules
pass through a gap, the molecules are kept in a tight bunch. We use this synchrotron to study collisions between stored ND3 molecules and beams of argon atoms. The advantage of using molecules stored in a synchrotron is two-fold: (i) The collision partners move in the same direction as the stored molecules, resulting in a low collision energy (down to 10cm-1); (ii) by storing molecules many roundtrips, the sensitivity to collisions is greatly enhanced.
Rather than trapping particles in a trap, as is common in atomic physics,
particles can also be stored in rings. We have developed a synchrotron for
low-energy neutral molecules composed of 40 hexapoles arranged in a circle.
By switching the voltages applied to the hexapoles every time the molecules
pass through a gap, the molecules are kept in a tight bunch. We use this synchrotron to study collisions between stored ND3 molecules and beams of argon atoms. The advantage of using molecules stored in a synchrotron is two-fold: (i) The collision partners move in the same direction as the stored molecules, resulting in a low collision energy (down to 10cm-1); (ii) by storing molecules many roundtrips, the sensitivity to collisions is greatly enhanced. Methyl alcohol is one of the simplest molecules that exhibits internal rotation; the methyl
(CH3) group rotates with respect to the alcohol (OH) group. In addition, the molecule rotates as a whole. Microwave
transitions that convert the internal rotation to overall rotation -- and vice versa --
are very sensitive to the proton-to-electron mass ratio. When the proton-to-electron mass ratio changes by a
certain fraction, the resulting fractional frequency change in methyl alcohol is up to 50 times this fraction.
This is an order of magnitude larger than the transitions used so far in searches for possible spatial of
temporal variations of the proton-to-electron mass ratio.
Methyl alcohol is one of the simplest molecules that exhibits internal rotation; the methyl
(CH3) group rotates with respect to the alcohol (OH) group. In addition, the molecule rotates as a whole. Microwave
transitions that convert the internal rotation to overall rotation -- and vice versa --
are very sensitive to the proton-to-electron mass ratio. When the proton-to-electron mass ratio changes by a
certain fraction, the resulting fractional frequency change in methyl alcohol is up to 50 times this fraction.
This is an order of magnitude larger than the transitions used so far in searches for possible spatial of
temporal variations of the proton-to-electron mass ratio.